Mycoplasma contamination in cell culture
|
Together with cross contamination, mycoplasma contamination is the worst problem we have in cell culture! Besides that, mycoplasmas are the most malicious microbiological contaminations we know. Why ? Because we can't see them in the light microscope, they don't float or really overgrow the cultures and this is why they don't kill our cultures quickly. That might sound rather nice but actually the consequence is that we don't easily realize that they are present. Many cultures are mycoplasma contaminated already for decades without us realizing it. And to even top that, they come with us to the lab each and every day as we carry them on our skin and mucosa. The worst thing about them, however, is that - for some strange reason - they seem to be able to convince us that only other people have mycoplasma contamination and that tests are superfluous as we would of course realize if we ever had a problem. You don't believe that ? Well, at present only around 30% of cell culture labs test for mycoplasmas! That this is not a good idea, what effects mycoplasma contamination has on your culture and how to test is what we explain on these pages. |
 |
The following topics are covered on this page:
Incidence of mycoplasma contamination
Mycoplasma detection and test kits
Species found in cell cultures
Mycoplasma classification
|
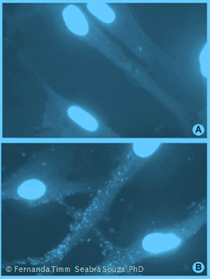
Mycoplasma are bacteria and belong to the class Mollicutes (soft skin) as - naturally they do not possess a cell wall. Originally, they were thought to be viruses and later were named PPLO (pleuropneumonia-like organisms). Today this term is only rarely used. As Mycoplasma species have no cell wall they are constitutively resistant to all cell culture antibiotics that inhibit cell wall synthesis, like penicillin, streptomycin and gentamycin. In addition, the lack of a cell wall also allows mycoplasma species to pass through 0,2µm membrane filters. The exerted pressure squeezes the mycoplasma cells through the pores even though they are usually larger than the 0,2µm.In the image you see a mycoplasma cell on the left and a normal bacterial cell with a red cell wall on the right. |
 |
Effects on cell culture
All mycoplasma species are parasites! This means they have to live on or in other cells and they steal nutrients and other metabolites from cells and cell culture medium. Additionally, they secrete toxins that harm or poison the cells. Below you find a list of effects described in literature:
|
Incidences of mycoplasma contamination in cell cultures
International cell banks, like DSMZ, ECACC or ATCC usually test their cultures on a regular basis. Some of them in addition to some companies publish the results of ring tests. These usually are a good indictaor for the general incidence of mycoplasma contaminations.
|
Sources of mycoplasma contamination in cell culture
Mycoplasma contamination can be categorized into two main categories: a) direct and b) indirect contamination. A direct contamination means that accidentally contaminated reagents are added to cells because they have not been tested (FCS, growth factors, coating proteins). Indirect contamination is mostly due to mistakes in aseptic technique. Thereby, skin germs or contamination from other cultures are introduced into a cell culture.
|
Mycoplasma detection methods
Mycoplasma detection can be achieved by several different test methods:
|
Among these methods are two that are most recommendable due to price, ease, specificity and sensitivity and these are the DAPI stain and PCR. A DAPI or Hoechst stain is easy and fast even though it is harder to read-out. The PCR test is the state-of-the-art technique as it combines - in the best case - ease of performance, speed, price, sensitivity and specificity. Below we have crudely outlined how - in a research lab - you can perform mycoplasma tests using one of these two methods.
PCR detection
In the PCR example, we show how to test an FCS batch that was newly purchased. First you prepare a known clean cell culture. To this, you add medium containing the new FCS batch. Now you grow and passage the cells (best without antibiotics) for at least a week to give the potential mycoplasma contamination time to grow. Longer culture periods would be safer. Then, you take a media sample from this culture. After boiling for approximately, 5min the sample is ready for testing using a PCR kit like the ones listed below. After 3-4 hours you get the result as bands on a gel.
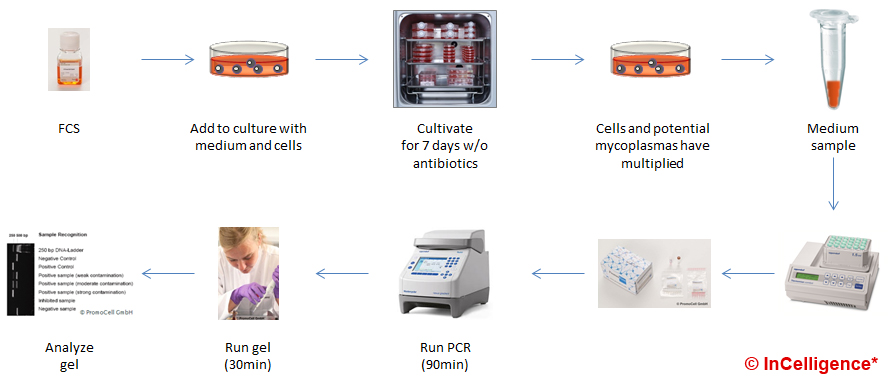
When performing a PCR detection of mycoplasmas in a GMP production surrounding, this very crude and basic protocol has to be changed in several aspects and controls have to be included. This is why research level kits have a true sensitivity of roughly 1.000 CFU/mL while GMP final product testing uses tests validated to have a sensitivity of 10 CFU/mL for all relevant species.
Detection by DAPI stain
The second example outlines the DAPI stain. You start off as described above with adding the test sample to a clean culture and culturing for at least 7 days. Then. you discard the medium, you wash and fix the cells with formaldehyde and stain them with DAPI or Hoechst solution. Now you can read out the result using a fluorescence microscope under the UV setting. As this method is much less sensitive it is better to prolong the culture period to 14-21 days in order to be really sure.

To use this test in a GMP production surrounding not much more than the cultivation period has to be changed. Of course controls have to be included.
Mycoplasma detection and test kits
| Supplier | Method/DL/GMP | Species / Test result type | Price | Remarks: Research kits |
| Example video |
PCR |
28 (yes/no answer) =
|
<6,63 |
Positive control: yes
In our MOOC, we have used this kit to demonstrate the method of PCR in a Video . |
| Minerva |
PCR-Gel <1000-2500 CFU GMP: - |
28 (yes/no answer) =
|
<10,34 |
Positive control: yes |
| Sigma |
PCR-Gel <1000-2500 CFU GMP: - |
19 (yes/no answer) =
|
<9,96 | Positive control: yes Internal control: yes Simplified kit as described above with lyophilized reagents in tubes. LookOut® |
| Supplier | Method/DL/GMP | Species / Test result type | Price | Remarks: No internal control |
| R&D Systems |
Hybridization- ELISA ~15 - 2500 CFU GMP: - |
8 (yes/no answer) =
|
~4 | Positive control: yes Internal control: no With this kit you detect mycoplasma rRNA in a colorimetric ELISA. The assay cross reacts with Lactobacillus and therefore, is not mycoplasma-specific. In addition the sensitivity is lower. MycoProbe® |
| Agilent |
PCR-Gel n.a. GMP: - |
Description states "most" (yes/no answer, 5 identified) | ~9,8 | Positive control: yes Internal control: no This kit only contains the primers. No polymerase or master mix are includ. After a PCR, 5 species can be identified in a restriction digest run on a gel. The product information does not state what is meant with "most" species. Inquire and compare to the lists below! The internal control provided is a mycoplasma DNA and therefore only serves as a PCR control. Mycoplasma Plus Primer set |
| Supplier | Method/DL/GMP | Species / Test result type | Price | Remarks: Research kits |
| Greiner BioOne |
PCR- Mikroarray <10 CFU GMP: + |
all known species (yes/no answer), (EuPharm 9 of 7-9) 40 (identifiziert) |
>560 |
Positive control: yes |
| Roche |
PCR-Gel <10 CFU GMP: + |
all known species (yes/no answer), (EuPharm ? of 7-9) | >500 |
Positive control: yes |
* Price is price per sample for a kit size ~ 25 samples.
Mycoplasma species listed in the EuPharm.:
The European Pharmacopoiea precisely lists how to test for mycoplasma contamination in sterile products and which species to use for validation and verification. Depending on what you test, the detection limit has to be 10-100 CFU per mL. Depending on the product, one of these control species is run as a positive control in all batch tests. Purified mycoplasma DNA should never be used as a GMP positive control as it does not realistically reflect the results obtained with intact mycoplasma as a positive control.
- A. laidlawii
- M. fermentans
- M. hyorhinis (where cell-culture enrichment is used, a fastidious strain such as ATCC 29052 is included)
- M. orale
- M. pneumoniae or M. gallisepticum
- M. synoviae (where there is use of or exposure to avian material during production)
- Mycoplasma arginini
- Spiroplasma citri (where there is use of or exposure to insect or plant material during production).
The most important and relevant species for culture
There are several test systems and test kits on the market. Several of these detect only a limited number of species. Make sure that the species below are in the detection range (specificity and sensitivity) of the kit you purchase. There are several kits that are cheap and have a wide range of detection.
- M. orale
- M. hyorhinis
- M. arginini
- M. fermentans
- M. salivarium
- M. hominis




